We have discussed bispecific antibodies (BsAbs) in this blog before, and we would like to announce a new publication in Bioprocess International on this topic. Teneobio, Inc., a biotechnology company in Menlo Park, CA, is developing a BsAb as a potential therapeutic for the treatment of cancer. Chamow & Associates is assisting Teneobio to produce this novel therapy for clinical testing.
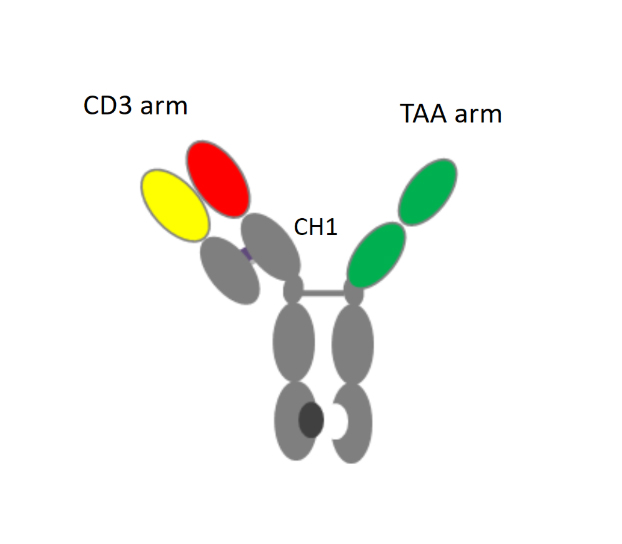
This IgG4 BsAb is directed against a tumor associated antigen (TAA) in combination with CD3 (CD3-TAA). In designing the manufacturing process, Teneobio scientists found that this BsAb tends to aggregate when exposed to low pH. Thus, use of Protein A with elution at low pH was problematic and stimulated us to investigate affinity resins that could serve as suitable alternatives to Protein A. In contrast to typical mAbs, Teneobio’s BsAb is a trimer. The CD3 arm contains both fully human heavy and light chain, while the TAA arm, derived from UniRat™ technology, consists only of a human heavy chain (with VH domains fused directly into hinge-CH2-CH3). Directed against two different antigens, it functions as a heterodimer. Only the heterodimeric product (and not the product-related TAA homodimer) contains a CH1 domain of human heavy chain. Two criteria guided our search for an alternative capture media: (a) the ability to elute product under mild conditions, and (b) selectivity for the heterodimeric product over the TAA homodimer as a process impurity. CaptureSelect CH1-XL, commercially available from ThermoFisher, is an affinity resin that binds specifically to the CH1 domain on the heavy chain of human IgG with the benefits of a robust and high‑quality affinity matrix provided by a 13 kDa llama heavy chain antibody fragment.
The work to design this novel capture step for purification of CD3-TAA BsAb is described by Dr. Chamow and colleagues in an article entitled “Capture of CH1-Containing Bispecific Antibodies: Evaluating an Alternative to Protein A” that appears in the May 2020 issue of Bioprocess International. View the full article on our Publications page here: http://chamowassociates.com/wp-content/uploads/2020/05/Chamow_Capture-of-BsAb_2020.pdf.
A preliminary report of this work was presented by Dr. Chamow at the 2018 BioProduction Congress in Dublin, Ireland. Chamow & Associates assists Teneobio and other biotechnology companies to develop biologics for clinical testing and welcomes your inquiry.